Naltrexone, an opiate treatment medication, is used to help patients make the transition from illicit opiate use to a drug-free life. Patients in naltrexone treatment are first detoxified from their dependence on opiates and then take thrice-weekly doses of naltrexone and participate in weekly group therapy sessions.
The medication provides a safety net for patients because it blocks the euphoric effects they normally would feel if they slip and use heroin or any other opiate. As a result, even relapse, which is common in addiction treatment, may have a therapeutic effect as repeated failure to get high may eventually break the neurobiological and behavioral links between taking drugs and the rewards that lead patients to resume regular drug use. With successful naltrexone treatment, slips to drug use become less frequent, the medication is discontinued, and patients continue behavioral treatment if needed.
Naltrexone treatment has been successful mainly with patients who are highly motivated to stop using opiates. Such patients include health care professionals who must stop using opiates to retain their licenses to practice medicine and individuals subject to criminal justice sanctions for relapse to illicit opiate use. The severe penalties that these patients would incur if they fail treatment enable them to overcome naltrexone's main drawback: It eliminates the powerful rewarding effects of opiates without any replacement to help patients cope with lingering effects of withdrawal.
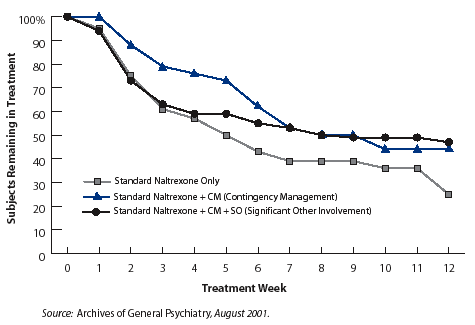 Voucher Incentives Increase Retention In Naltrexone Treatment In a study with 127 heroin-addicted patients receiving naltrexone therapy, the 12-week dropout rate was about 50 percent among those in two groups that received voucher-based contingency management, and about 75 percent among those who did not.
Voucher Incentives Increase Retention In Naltrexone Treatment In a study with 127 heroin-addicted patients receiving naltrexone therapy, the 12-week dropout rate was about 50 percent among those in two groups that received voucher-based contingency management, and about 75 percent among those who did not.Naltrexone's lack of a reinforcing effect has made it an unattractive treatment option for other patients who lack a strong external incentive to stop using drugs and do not want to go through detoxification and withdrawal from opiates. Most of these patients opt for treatment with medications such as LAAM and methadone, both of which help them to cope with the absence of the intense and rapid high that they are accustomed to getting from heroin by replacing it with a more moderate, stabilizing effect that can help them to maintain a nonaddicted lifestyle.
Despite its limited clinical use, naltrexone has many qualities that make it an attractive option for treating a broader range of opiate-dependent patients. It is not addicting, has few adverse effects, can be prescribed without concerns about diversion to the illicit drug market, and is not subject to the restrictive regulatory requirements that limit the use of methadone and LAAM to specialized clinics. Thus, like the recently approved opiate treatment medication buprenorphine, naltrexone can be administered in many settings, including private physicians' offices, making it more attractive to individuals who are reluctant to enter clinics.
Naltrexone's desirable therapeutic traits have continued to spark interest in finding new ways to expand its usefulness and application in practice. Two recent studies show that adjunctive behavioral and new pharmacological approaches may help to increase naltrexone's effectiveness for a wider range of opiate-addicted patients.
Voucher Reinforcement Increases Naltrexone's Effectiveness
A NIDA-supported treatment study that rewarded heroin-dependent patients with vouchers whenever they took their naltrexone or tested negative for drug use has found that this basic behavioral reinforcement approach achieved significantly better results than standard naltrexone treatment alone in keeping patients in treatment longer, having them complete treatment, and reducing their opiate use.
"A significant boost in treatment adherence was achieved not with highly motivated patient groups that have generally responded well to naltrexone treatment, but with predominantly unemployed 'street addicts,' most of whom had a history of extensive involvement with drug abuse treatment and the legal system," says Dr. Dorynne Czechowicz of NIDA's Division of Treatment Research and Development. She also maintains that the results are promising for expanding the types of patients who would benefit from naltrexone treatment.
The 12-week study, led by Dr. Kathleen Carroll of the Yale University School of Medicine, randomly assigned 127 recently detoxified opioid-dependent patients to 1 of 3 treatment conditions: standard treatment with naltrexone 3 times a week; standard naltrexone treatment plus a behavioral reinforcement approach called contingency management (CM); or standard naltrexone treatment and CM plus involvement of a significant other (SO) in up to 6 family counseling sessions. SO treatment was added to CM for patients in the third group to test the idea that encouragement and positive reinforcement from a significant other might help patients cope with any protracted drug withdrawal symptoms and remain in treatment longer. Patients in all three groups participated in weekly cognitive-behavioral group counseling sessions.
Patients in the CM groups could earn vouchers, which they could exchange for goods and services, in separate tracks for naltrexone compliance or drug-free tests. In each track, the voucher value started at $0.80, escalated in $0.40 increments for continuous compliance or abstinence, and were reset to the starting point for each failure to take the medication or pass a drug test. Over the course of the study, patients in the CM groups earned an average of $189 in vouchers out of the maximum $561 that could be earned for perfect medication compliance and all negative drug tests.
The researchers found that on average, patients in the two CM groups stayed in treatment 7.4 weeks, significantly longer than the 5.6 weeks for those in standard treatment. A much higher percentage of CM patients also completed the full 12-week treatment period--47 percent of CM plus SO patients, 42.9 percent in the CM group, and 25.6 percent of patients in the standard treatment group. These retention rates with CM added to standard treatment also compare favorably with rates achieved in previous studies of standard naltrexone treatment, which have reported that 60 to 70 percent of patients dropped out of treatment over a 12-week period, Dr. Carroll notes.
Patients in the CM groups also had significantly better treatment outcomes than those in the standard naltrexone group--more days of abstinence, longer periods of continuous abstinence, more opiate-free tests, and a higher percentage of drug-free specimens. Additional analyses suggested CM patients made greater reductions than standard treatment patients in the frequency with which they used opiates as the study progressed. Thus, 100 percent of patients reported weekly opioid use at the beginning of the study, but fewer than 10 percent of those who completed treatment reported weekly use over the last 4 weeks of the study. Although adding SO to CM did not improve most treatment outcomes, further analysis suggested it did produce a significant reduction in family problems over time.
"Our study shows you can really bump up medication compliance and outcomes with very simple behavioral interventions," Dr. Carroll says. "It doesn't take much effort or cost for treatment programs to do this, particularly if you look at the potential savings from keeping patients in treatment longer where they can learn how not to be drug users."
Source
- Carroll, K.M., et al. Targeting behavioral therapies to enhance naltrexone treatment of opioid dependence: Efficacy of contingency management and significant other involvement. Archives of General Psychiatry 58(8):755-761, 2001. [Abstract]