Details
Contact
United States
Meeting Summary
NIDA’s Neuroscience Consortium organized the 10th annual Frontiers in Addiction Research Mini-Convention held at the 2011 Society for Neuroscience Meeting on November 11, 2011, in Washington, DC. The mini-convention included sessions on autism, addiction, and the role of the MeCP2 gene in mediating response to drugs; synapse organization, and plasticity and its relationship to drug addiction; using optogenetics to shed light on the neural mechanisms of addiction; and the neurobiology of behavioral and emotional regulation/dysregulation.
- Interviews with NIDA scientists at the 2011 Society for Neuroscience meeting
-
Session Summaries
- Autism, Addiction and MeCP2
-
Overview
The MeCP2 protein binds methylated cytosine residues in DNA. In 1999, the MeCP2 gene was discovered to be altered in many cases of the autism spectrum disorder Rett syndrome. Since then, we have learned much more about the functions of MeCP2 in the mammalian nervous system, including an unexpected role for MeCP2 in mediating responses to certain drugs of abuse. As a whole, this mini-symposium will provide insights into the possible roles of MeCP2 in neurodevelopmental and substance abuse disorders.
The Evolving Role of MeCP2 in Human Neurodevelopmental Disorders
Janine M. LaSalle, Ph.D. UC Davis School of Medicine
The combination of observable traits, also referred to as phenotypes, can be influenced by gene expression and environmental factors, including exposure to drugs. Dr. LaSalle began her presentation by discussing the genes related to neurodevelopmental disorders and what they tell us about epigenetics’ role in the development of the social brain. MeCP2, a binding protein known for regulating gene expression, produces one of the essential “readers” of the DNA methylation code, the first layer of epigenetic information in developing neurons. MeCP2 regulates the genes involved in changes to neural activity and growth, as well as the genes that control anxiety and stress response. These genetic mechanisms affect a wide range of neurodevelopmental disorders, particularly autism spectrum disorders. When MeCP2 is deficient, it causes neuronal nuclei to get smaller and chromosome structure to change.
Dr. LaSalle and her team of researchers developed a model that allowed them to map the genomic locations of partially methylated domains in human neuronal cells. This mapping helped them determine how much of a role DNA methylation plays in the neuronal nuclear changes that occur during development. These results indicate that the level of DNA methylation and MeCP2 is critical to the development of the human brain, as reduced DNA methylation caused by exposure of MeCP2 mutant female mice to a common chemical flame retardant resulted in a decrease in sociability in their female offspring. This is a classic behavioral characteristic among individuals with autism spectrum disorders and in individuals known to abuse drugs.
Cocaine-Induced Chromatin Remodeling of BDNF Transcription in the mPFC
Ghazaleh Sadri-Vakili, Ph.D., MassGeneral Institute for Neurodegenerative Disease, Massachusetts General Hospital; Harvard Medical School
 Download the Presentation (PDF, 1.1MB)
Download the Presentation (PDF, 1.1MB)The effects of cocaine are not limited to individuals exposed to cocaine. They can be transmitted to the user’s offspring through the germ line, even when those generations were never exposed to cocaine, suggesting that the population at risk may be larger than scientists originally estimated. Dr. Sadri-Vakili explained what researchers know about the contribution of epigenetic factors to the risk of cocaine abuse. Using animal models of addiction, the research team demonstrated that drugs cause epigenetic alterations in gene expression that can influence brain development and behavior.
Cocaine-induced alterations in the brain are characterized by behavioral, cellular, and molecular events, including alterations in cocaine self-administration and locomotor sensitization to cocaine, increased BDNF expression, increased histone acetylation, and decreased MeCP2 binding. Dr. Sadri-Vakili and her team of researchers used paternal and maternal animal model to characterize these events across multiple generations. In the first model, male rats self-administered cocaine for 60 days, mated with drug-naïve females, and then behavior and molecular traits were examined in the F1 generation at adulthood. In the second model, pregnant mice received twice-daily injections of cocaine until birth and behavioral and molecular traits of the F1 and F2 generations were examined at development and adulthood. Results showed that male offspring of rodents exposed to cocaine demonstrated a significant change in behavior, brain development, and gene expression in both models in the F1 and F2 generations. More specifically, the male offspring were less sensitive to the rewarding effects of cocaine that was marked by increased levels of BDNF protein and mRNA in the prefrontal cortex.
MeCP2 and Psychostimulant-Induced Behavioral Adaptations
Anne E. West, M.D., Ph.D., Duke University Medical Center
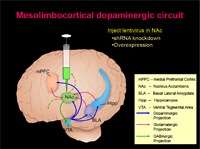
Psychostimulants like cocaine and amphetamine very rapidly enter the brain and increase levels of dopamine. Over the long term, studies show that exposure to psychostimulants results in locomotor sensitization, memory challenges and eventually addiction. However these behavioral changes require chronic exposure to psychostimulants, suggesting that slower biochemical processes may underlie these behavioral changes. Many studies suggest that neural plasticity mechanisms, including changes in the way genetic information is copied into new RNA molecules, are essential in this transition to addiction. Dr. West discussed research measuring the epigenetic mechanisms of chromatin regulation in response to repeated exposure to psychostimulants and its role in these behavioral adaptations.
Dr. West and her team of researchers used a mouse model to determine whether psychostimulants act via the MeCP2 protein, which modulates gene transcription, to influence long-lasting behavioral changes. Her results showed that when the MeCP2 is mutated in a way that prevents it from being regulated by psychostimulants, that mice experience more rapid locomotor sensitization to amphetamine and demonstrate increased sensitivity to the rewarding effects of cocaine.
These results suggest that MeCP2 limits the rewarding and reinforcing properties of psychostimulants like amphetamine and cocaine. Dr. West explored the implications of this data and what they tell scientists about the molecular and cellular mechanisms that influence the development of addictive behaviors.
- Using Optogenetics to Shed Light on the Neural Mechanisms of Addiction
-
Overview
Within the last few years, optogenetic techniques within the neuroscience field have been providing important insights into the underlying neural mechanisms of drug addiction and other brain disorders. Optogenetic methods selectively target and control the activity of specific neurons that have been modified to express either light-sensitive cation channels that can depolarize the neuron, or chloride pumps that can produce hyperpolarization. Optogenetics has the advantage of selectively targeting neurons for stimulation or inhibition. This selective targeting can be accomplished using more traditional electrical or pharmacological neuron-modulatory techniques. This session provided a brief overview of this technology, with additional presentations focusing on the use of optogenetics in exploring the neuronal mechanisms of drug addiction.
Activation of Ventral Tegmental Area GABAergic Neurons Disrupts Reward Consumption
Garret D. Stuber, Ph.D., University of North Carolina at Chapel Hill
The ventral tegmental area (VTA) is a brain structure containing groups of neurons essential for the expression of behaviors related to addiction and other neuropsychiatric illnesses. Some of these neuron groups work together to control behavioral responses; however, it has been difficult for researchers to precisely test these mechanisms in a living organism.
Dopaminergic (DAergic) neurons, those that contain dopamine—the neurotransmitter that helps control the brain’s reward and pleasure centers, are combined with GABAergic neurons in the VTA. Although 30-40 percent of neurons in the VTA are GABAergic, their function is not yet understood. Dr. Stuber and his team of researchers used optogenetic strategies to study how modulating the activity of GABAergic neurons in the VTA influences reward-seeking behavior by decreasing or increasing levels of dopamine in the brain.
To examine how reward-related behaviors are influenced by the activation of GABAergic neurons, the team used a light stimulus to predict the delivery of a sucrose reward to adult mice. They then activated GABAergic neurons timelocked either to a reward-predictive cue or to reward consumption. Results showed that optogenetic activation of neurons timelocked to reward-predictive cues did not alter reward-seeking during the cue presentation or during reward consumption. However, optogenetic activation did significantly disrupt reward consumption in GABAergic neurons timelocked to reward delivery. Results also showed that the firing rates of DAergic neurons in the VTA were reduced when GABAergic neurons were activated. This presentation demonstrated that by modulating specific neuron groups in the VTA, researchers can alter the brain’s response to rewards and pleasure.
Genetic and Optogenetic Manipulation of Medium Spiny Neuron Subtypes
Mary Kay Lobo, Ph.D., University of Maryland School of Medicine
The nucleus accumbens (NAc) plays a crucial role in mediating the rewarding effects of drugs of abuse, but until recently, little was known about the pathways and processes of the NAc’s medium spiny neurons (MSNs). MSNs account for more than 95 percent of all NAc neurons and they are divided into two subtypes based on their expression of specific genes including dopamine receptors 1 and 2 (D1 and D2) and the regions they project to in the brain. To understand the role of MSNs in cocaine-reward behaviors, Dr. Lobo and her team of researchers used genetic mouse models to manipulate BDNF signaling, which is involved in the regulation of stress response and the biology of mood and addiction disorders. They specifically deleted TrkB, the BDNF receptor, from D1 containing vs. D2 containing MSNs.
The team then used optogenetic methods to manipulate neuronal firing in each MSN for comparison with TrkB deletion. Data revealed that loss of TrKB and turning on neuronal firing selectively in each MSN resulted in opposing behavioral responses to cocaine. Deletion of TrKB in D1 MSNs and activation of D1 MSNs with optogenetics increased the effects of cocaine reward, while deletion of TrKB in D2 MSNs and activation of D2 MSNs reduced the effects of cocaine reward. Dr. Lobo explained that her team’s findings were consistent with current models of opposing functions of the two MSN subtypes, with respect to other behaviors including motor control. Her presentation provided insight into how neuronal activity is controlled on the molecular level and how dopamine receptor cell types contribute to reward triggers in the brain.
Decreased Excitability of Prelimbic Pyramidal Neurons Induced by Extended Cocaine Self-Administration Contributes To Compulsive Drug Seeking
Billy T. Chen, Ph.D., NIDA Intramural Research Program
 Download the Presentation (PDF, 946Kb)
Download the Presentation (PDF, 946Kb)Research shows that different brain regions are involved in an individual’s transition to drug abuse. As the ability of neurons to compensate for injury and disease fluctuates within these regions, drug seeking behaviors can become more compulsive. Previous studies suggest that compulsive drug-seeking may result from inadequate functioning of the decision-making centers in the prefrontal cortex. This presentation expanded on those previous studies, as it explored whether long-term cocaine self-administration caused the decision making parts of the prefrontal cortex to fail and neuronal activity to change, thereby influencing compulsive drug use.
To test this hypothesis, Dr. Chen and his research team trained adult male rats to self-administer cocaine on a seek-take chain schedule for about 2 months. After this period, they were exposed to foot shock punishment during self-administration. Researchers observed both a shock-resistant group (continued to self-administer despite shocks) and a shock-sensitive group (suspended all cocaine-seeking when shocked) before performing ex vivo studies of deep-layer pyramidal neurons in the prelimbic area. Results showed that neurons in the prelimbic area of cocaine-experienced rats showed significant decreases in membrane excitability compared to cocaine-naïve rats. Furthermore, this effect was more pronounced in shock-resistant rats than in shock-sensitive rats.
The research team also used in vivo optogenetic stimulation to rescue cocaine-induced hypoactivity in the prelimbic area in the shock-resistant rats. Dr. Chen’s findings showed that prolonged cocaine self-administration decreased excitability in prelimbic neurons, which may contribute to compulsive cocaine-seeking behaviors.
- Neurobiology of Behavioral and Emotional Regulation/Dysregulation
-
Overview
Genetic, neurobiological, and prevention research suggest that a generalized inability to control one’s behavior and emotions predisposes an individual to substance use and other psychiatric disorders. However, knowledge of a common liability factor does not necessarily shed light on which disorders will develop, or on what sorts of treatment will be most effective. Once a specific disorder or comorbidity develops, disease-specific neuroadaptations and epigenetic modifications may come into play, induced by exposure to abused substances or other environmental factors. This symposium will present recent studies that investigate the neural substrates of behavioral regulation and new approaches to testing the usefulness of a construct of a “dysregulation syndrome” for understanding the etiology of specific disorders and how to treat them.
Neural Mechanisms Linking Behavioral Dysregulation in Substance Abuse, Psychopathology, and Stress
Anthony A. Grace, Ph.D., University of Pittsburgh
 Download the Presentation (PDF, 2.2MB)
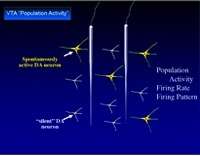
Download the Presentation (PDF, 2.2MB)External stressors do not create identical responses within the nervous system. This is evident in the adaptive reactions to stress that benefit survival by motivating us to avoid dangerous situations, as well as the maladaptive reactions that lead to drug abuse, post-traumatic stress disorder, and depression. Dr. Grace presented research suggesting that stressors that vary in intensity and duration can affect the dopamine (DA) system in different ways. He began by discussing the types of stressors—noxious, physiological, and psychological—and the unique ways they affect our nervous system. Dr. Grace and his research team used footshocks, restraint, and exposure to chronic cold to simulate the effects of unique brain stressors.
Dr. Grace explained the processes used to observe the activity of DA neurons in the ventral tegmental area (VTA) and responses to stressors, including counting the number of neurons firing spontaneously, measuring the average firing rate and neuronal patterns, and assessing amphetamine-induced locomotor activity. Results showed that intense noxious or psychological stressors activated the DA system and increased behavioral response to amphetamine. Dr. Grace explained that this condition illustrated the type of response that might occur with post-traumatic stress disorder or anxiety disorders. In contrast, mild inescapable stressors depressed the reward-related DA neurons, which is consistent with an individual’s inability to gain pleasure from enjoyable experiences in cases of depression. The uniqueness of these stress responses lends to unique impacts on an individual’s risk for abusing drugs that act on the DA system.
An Evolutionary Perspective on Behavioral Syndromes: Insights from Whole Genome Expression Data
Alison M. Bell, Ph.D., University of Illinois, Champaign-Urbana
 Download the Presentation (PDF, 1MB)
Download the Presentation (PDF, 1MB)External stressors do not create identical responses within the nervous system. This is evident in the adaptive reactions to stress that benefit survival by motivating us to avoid dangerous situations, as well as the maladaptive reactions that lead to drug abuse, post-traumatic stress disorder, and depression. Dr. Grace presented research suggesting that stressors that vary in intensity and duration can affect the dopamine (DA) system in different ways. He began by discussing the types of stressors—noxious, physiological, and psychological—and the unique ways they affect our nervous system. Dr. Grace and his research team used footshocks, restraint, and exposure to chronic cold to simulate the effects of unique brain stressors.
The Neurocircuitry of Emotion Regulation: The Control of Fear and Implications for Treatment of PTSD and Comorbid Disorders
Elizabeth A. Phelps, Ph.D., New York University
There are several brain regions involved in the regulation of emotion in human beings, including the prefrontal cortex, hippocampus, and amygdala. Dr. Phelps and her team of researchers used animal model research and observations of similar behavior in human beings to determine how we can control and diminish responses to fear through extinction, reconsolidation, and emotional regulation. Comparisons of research on fear conditioning in rats and humans illustrate that both species show similar responses, including increases in blood pressure, heart rate, and respiration, and the release of stress hormones. This presentation discussed coping techniques that can influence how humans regulate emotions and techniques for altering emotional responses, including disrupting the neurological/psychological process that occurs when long-term memories are drawn out, strengthened, or re-imprinted.
Extinction takes places when repeated exposure to a learned fear stimulus with no aversive consequences results in new learning that the conditioned stimulus is now safe. It forms the basis for exposure therapy treatment of phobias. Animal models show that activity in the ventral medial prefrontal cortex (vmPFC) is necessary for extinction to occur and that the amygdala plays a key role in its success. Dr. Phelps explained how cognitive therapists use emotional regulation to help patients reassess whether to associate a stimulus with a negative affect into neutral or positive emotional terms. In this case, the vmPFC controls fear by inhibiting the amygdala, which controls expression and helps with the formation and storage of memories associated with emotional events. She also explained study results relating to the reconsolidation of a conditioned fear response and the retrieval–extinction procedure, known to act on neural circuits in the brain to prevent drug craving and relapse.