Twenty-five years ago it would have been almost impossible to imagine a treatment for opiate addiction that could be prescribed in a physician's office, picked up at a pharmacy, and taken at home. But that scenario has been achieved after a quarter-century of collaborative effort—and the overcoming of several barriers—by NIDA's medication development program and Reckitt Benckiser Pharmaceuticals, Inc.
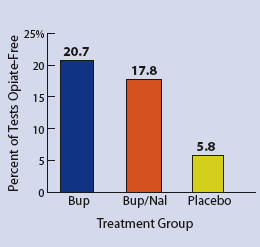 Buprenorphine and Buprenorphine/Naloxone Help Patients Quit Opiate Abuse. Patients undergoing treatment for opiate addiction who received buprenorphine or buprenorphine plus naloxone were more likely to test negative for opiate abuse than patients given placebo. Craving for opiates also was reduced in the two treatment groups.
Buprenorphine and Buprenorphine/Naloxone Help Patients Quit Opiate Abuse. Patients undergoing treatment for opiate addiction who received buprenorphine or buprenorphine plus naloxone were more likely to test negative for opiate abuse than patients given placebo. Craving for opiates also was reduced in the two treatment groups.Dr. Don Jasinski, a scientist at NIDA's Intramural Research Program (IRP), was the first to recognize the characteristics of buprenorphine— developed in the 1970s as an injectable pain medication—as useful for addiction treatment. He led the initial 1978 study demonstrating the drug's effectiveness and its acceptability to patients as a treatment for opiate dependence.
Early on, NIDA scientists realized that medications for addiction not only had to be safe and efficacious, but also had to be available in a form that would be practical for therapeutic use over the long term. NIDA worked with Reckitt Benckiser (then Reckitt & Colman) to develop noninjectable formulations of buprenorphine; by 1990, Dr. Ed Johnson and colleagues at the IRP demonstrated that a solution form of the drug administered under the tongue was safe, effective, and acceptable to patients as an opiate dependence treatment.
As with any opioid, however, there were concerns about buprenorphine diversion and the potential for abuse. NIDA again collaborated with the manufacturer, and by the mid-1990's, developed a combination tablet of buprenorphine and naloxone that would minimize the potential for abuse—a development that put the vision of take-home treatment for opiate dependence within reach. In the next decade, scientists at NIDA and Reckitt Benckiser conducted clinical trials with more than 2,400 patients that established buprenorphine's safety and efficacy in treating opiate dependence. And finally, a NIDA-funded collaborative clinical trial, codirected by Dr. Paul Fudala of the Veterans Affairs Medical Center and the University of Pennsylvania in Philadelphia, established the safety and effectiveness of the buprenorphine-naloxone combination as a prescribed take-home treatment. Data from this study and two other pivotal trials formed the basis for the U.S. Food and Drug Administration's (FDA's) approval of buprenorphine and the combination medication in 2002.
"People at NIDA knew of the great need to move opiate addiction treatment from the traditional clinic settings to individual physicians' offices. But we had to address concerns about diversion and unprescribed use. Drs. Jasinski, Johnson, and Fudala deserve a great deal of credit for their contributions to this collaborative achievement—a safe and effective take-home treatment with minimal likelihood for abuse," says Dr. Frank Vocci, director of NIDA's Division of Treatment Research and Development.
Dr. Fudala's research, a nationwide study of 472 opiate-addicted men and women, was codirected by Dr. T. Peter Bridge, then of NIDA, and was recently published. The study confirmed that the efficacy and safety of the combined therapy are equivalent to those of buprenorphine alone and superior to placebo. The combination reduces craving for and use of opiates, presents limited potential for abuse, and is suitable for office-based use, the investigators concluded.
Initial Treatment Outcomes
The study began with a double-blind phase in which 323 opiate-addicted individuals (ages 18 to 59) received one of three treatments for 4 weeks. One group of 109 patients received tablets totaling 16 mg buprenorphine and 4 mg naloxone; the second group (105 patients) received tablets totaling 16 mg buprenorphine only; and the third group (109 patients) received placebo tablets. All tablets were identical in appearance and taste. Patients reported to the clinics for dosing every weekday and took their medications home for weekends and holidays. Study patients and placebo patients also participated in up to 1 hour of individualized counseling per week. Opiate use was monitored through urine tests every Monday, Wednesday, and Friday.
The plan for the initial double-blind, 4-week arm of the study was to recruit 384 patients and provide each patient with 4 full weeks of therapy. However, recruitment was halted at 323 subjects because the patients receiving either medication clearly were doing better than the placebo patients. Both medication groups showed significant reductions in opiate use and craving and significant improvements in perceptions of overall health compared with those receiving placebo.
In the buprenorphine-naloxone group, the proportion of opiate-free tests was 17.8 percent; the buprenorphine group had 20.7 percent opiate-free tests; and the placebo group, 5.8 percent. The presence of cocaine, the nonopiate drug most commonly found in urine samples in this study, did not vary significantly among the three groups. Nor was there a noticeable difference among the treatment groups in drug-positive results for amphetamines, barbiturates, or methadone.
"The number of urine samples negative for drugs probably would have been higher if investigators had used the results to counsel patients. Such feedback is known to further reduce patients' drug use, but that information was not revealed to the researchers to prevent bias. The urine test results reflect higher use at the beginning of the study—when patients are ambivalent about treatment and in the grip of addiction. It's positive that opioid use decreased over the course of the study," says Dr. Vocci.
Patients in both medication groups also reported reduced craving for opiates. All groups showed the same average self-reported craving level before treatment—approximately 60 on a 100-point scale. By week 4 of the study, the average craving scores fell by half for both medication groups but did not change for the placebo group. Patients receiving medications reported greater improvement in overall health and well-being than those in the placebo group—perceptions confirmed by higher weekly clinician ratings of patients' overall health and well-being for the two buprenorphine-treated groups. Because both medications were clearly effective, the researchers halted the first phase of the study. Patients receiving placebo during this phase went on to receive buprenorphine-naloxone combination treatment in the second phase of the study.
Longer-term Efficacy
The goal of the study's second phase was to evaluate the safety of the combination tablet in more natural conditions and over a longer term, without the restrictions associated with the double-blind condition. In this open-label portion of the study, which lasted up to 52 weeks, all patients received the combination tablet. Weekly counseling was available along with a daily dose of up to 24 mg buprenorphine and 6 mg naloxone, tailored to each patient's individual response. The sublingual tablet was administered at the clinic each weekday for the first 2 weeks; after that, patients could take home up to a 10-day medication supply at the discretion of the investigator.
Of the 472 patients who began this phase of the study, 385 received at least 8 weeks of treatment, and 261 were treated for at least 6 months. Fourteen patients discontinued therapy because of adverse events, of which detoxification or withdrawal symptoms were the most common. Opiate-free urine samples in the open-label phase of the study ranged from 35.2 percent to 67.4 percent in multiple assessments. The overall rate of opiate use was lower than in the first phase of the study, but cocaine and benzodiazepine use remained relatively constant, the researchers reported.
The study concluded that the addition of naloxone to protect against illicit use of the treatment medication did not reduce the efficacy of buprenorphine.
"This new treatment option is historic," says Dr. Vocci. "Congress passed the Drug Abuse Treatment Act of 2000 so that buprenorphine products, and other Schedule III, IV, and V medications approved for opioid treatment by FDA, can be prescribed by qualified doctors for the treatment of opioid addiction. This represents a change to a level of prescribing privileges that American doctors have not had since the Harrison Narcotic Act of 1914."
Who Can Benefit
In the two years since the medication was approved, clinicians have gained an understanding of which patients are most likely to benefit from a take-home treatment option. Dr. Fudala cautions that buprenorphine is not likely to work well for every patient. Those less likely to benefit may include patients who require very high doses of methadone. Buprenorphine is a partial agonist, which means that in severely addicted people, it may not provide enough opiate agonist activity to treat them adequately.
Dr. Fudala says the combined agent may be especially useful for patients who do not have extremely high levels of addiction and for younger individuals, who typically have a shorter abuse history and may be using smaller amounts of an addictive substance. "We're seeing younger and younger heroin addicts these days," says Dr. Fudala. "It may be a good initial treatment for them, either as a medical detoxification or, if necessary, as a longer term treatment. We'll have a better understanding of this as we gain more experience." Another suitable population may be addicted professionals, including those in health care, who could be motivated to seek treatment in the privacy of a physician's office setting.
Buprenorphine's suitability for office-based prescribing is based on its pharmacologic profile. Like methadone, buprenorphine activates opiate receptors, but its effects level off as the patient takes higher and higher doses; this reduces the likelihood of dangerous side effects such as severe respiratory depression. The addition of naloxone reduces the potential for abuse by illicit injection: If a combination tablet is crushed and injected by a heroin-addicted individual in an attempt to intensify buprenorphine's euphoric effect, naloxone kicks in to induce the symptoms of opiate withdrawal. Finally, buprenorphine has a relatively long duration of action and causes comparatively mild withdrawal discomfort on cessation, affording flexibility in dosing regimens and a margin of convenience for patients and physicians.
As of March 2004, 3,951 U.S. physicians were eligible to prescribe buprenorphine. Of that group, 2,848 were granted waivers of a Federal requirement for previous experience in addiction medicine. This number is growing, according to Dr. Vocci. "We had estimated that about 6,000 physicians would eventually take the training and get the waiver. So we're at about 50 percent," he says. At this time, he notes, certified physicians are restricted to treating no more than 30 patients. In October 2005, 3 years from the approval of the new drug combination, the Department of Health and Human Services and the Drug Enforcement Administration will evaluate the program and possibly adjust the restrictions. The overall picture, however, is positive, says Dr. Vocci. "Very little diversion has been reported with this new combination," he says.
Source
- Fudala, P.J., et al. Office-based treatment of opiate addiction with a sublingual-tablet formulation of buprenorphine and naloxone. New England Journal of Medicine 349(10):949-958, 2003. [Abstract]